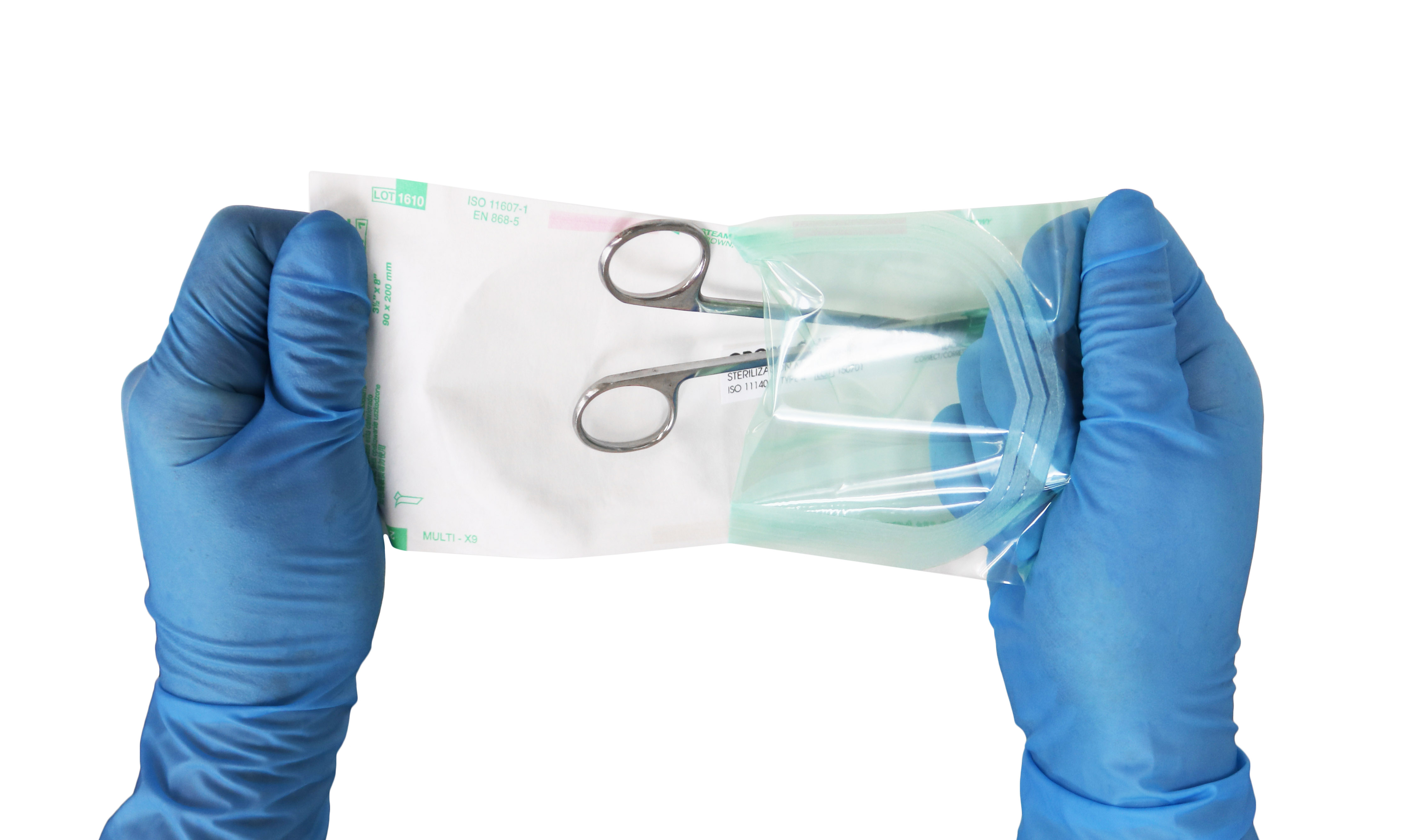
About ::Dr. Mary Ann Drosnock::
Dr. Mary Ann Drosnock is the Head of Clinical Affairs at Healthmark Industries, a Getinge company, where she leads clinical education, standards leadership, and infection prevention initiatives focused on improving the safety and effectiveness of medical device reprocessing.
She is internationally recognized for her expertise in endoscope reprocessing, microbiology, and infection prevention, and is a sought-after speaker at professional society meetings around the world. Dr. Drosnock currently serves as Co-chair of AAMI Working Group 40, the committee responsible for ST79, the foundational and most widely adopted standard in sterile processing. She also previously served as co-chair of the working group responsible for ST91, the national standard for flexible and semi-rigid endoscope processing.
Prior to joining Healthmark, Dr. Drosnock managed the Infection Control Program for Olympus, worked as a pharmaceutical microbiologist, and taught microbiology at the college level to healthcare professionals.
She holds advanced degrees in biology, quality and regulatory affairs, and health science, is certified in Infection Prevention and Endoscope Reprocessing, and is a Fellow of both APIC and AAMI.
Dr. Drosnock is widely respected for her ability to translate complex science, standards, and regulatory expectations into practical, real-world guidance for frontline teams.
HAVE A QUESTION?
WE ARE HERE TO HELP
Have a question about our products? Contact us today to speak directly to a Healthmark team member or shop our catalog now to request a inquiry